Chemical dominos
This project is very similar to the Rube Goldberg project I did as a freshman. The objective was to create a game and use chemical reactions to initiate a process by which players could play by. The project needed to include 1) Double Displacement/Double Replacement Reaction, 2) Single Displacement/Single Replacement Reaction, 3) Undergo a Physical Change, 4) Contain a Chemical Change, 5) Light up an LED or some other means of electrical light, 6) Must have a game board and playing pieces, 7) And needs to have safety and playing guidelines/instructions. Our project was displayed with a Steampunk theme, That is why I used many metal parts as well as gears and chains to create a more authentic look. As far as the background aesthetics, I wanted to make everything look shiny, or made out of metal hence the golden spray paint.
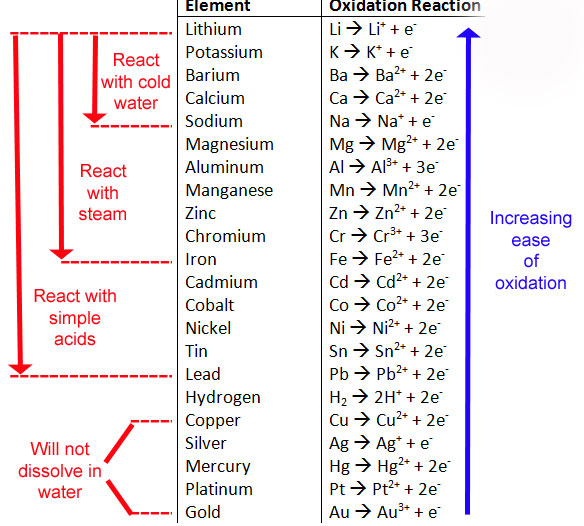
Single Replacement: C+AB --> A+CB An element reacts with a compound and takes the place of another element in that compound. Use the Activity Series of Metals (top) to predict the outcome.
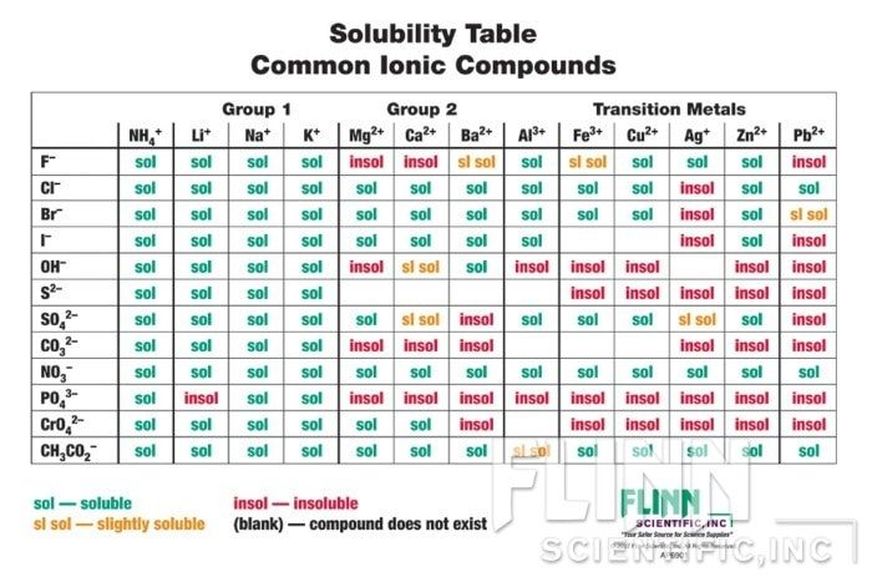
Double Displacement: AB+CD --> AD+CB Two compounds react, and the cation and anion of the reactants switch places forming two new compounds (Metathesis). Use the Solubility Chart (below) to predict the outcome.
Decomposition: ABC+D --> A+B+C+D Compounds break down into several elements or new compounds, often involving energy like heat, light, or electricity to break apart the bonds of the compounds.
Double Displacement: AB+CD --> AD+CB Two compounds react, and the cation and anion of the reactants switch places forming two new compounds (Metathesis). Use the Solubility Chart (below) to predict the outcome.
Decomposition: ABC+D --> A+B+C+D Compounds break down into several elements or new compounds, often involving energy like heat, light, or electricity to break apart the bonds of the compounds.
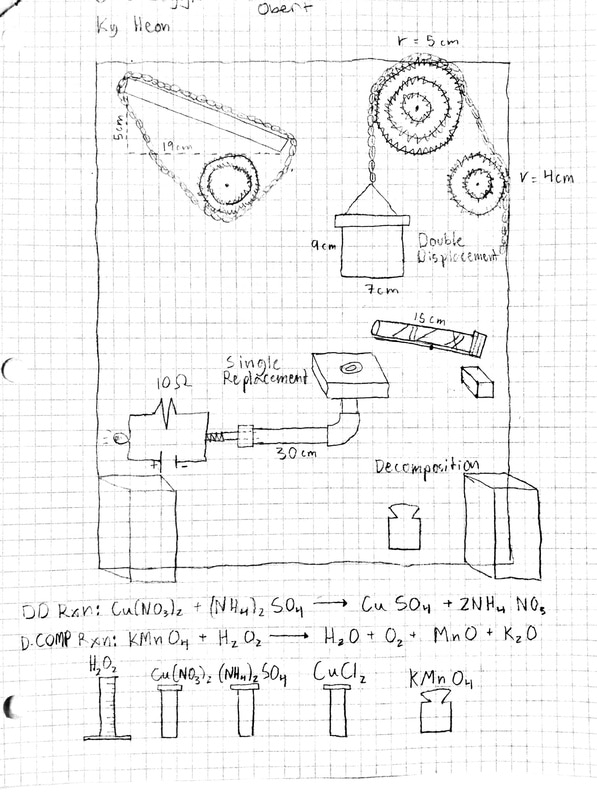
SCHEMATIC of Project
Materials:
-Flatwood
-Cardboard
-Nails & Screws
-2x4 Wood
-Gears
-Gear Chains
-Styrofoam Cup
-Laboratory Flask
-Metal Pipe
-Wires
-Washer
-Cardboard Spacers
-Aluminum Foil
-Scotch Tape
-Wooden Jump Rope Handles
-Incandescent Light & Circuitry
-C-Cell Alkaline Battery (3 Voltz)
-Metal Cogs
-Flatwood
-Cardboard
-Nails & Screws
-2x4 Wood
-Gears
-Gear Chains
-Styrofoam Cup
-Laboratory Flask
-Metal Pipe
-Wires
-Washer
-Cardboard Spacers
-Aluminum Foil
-Scotch Tape
-Wooden Jump Rope Handles
-Incandescent Light & Circuitry
-C-Cell Alkaline Battery (3 Voltz)
-Metal Cogs
Chemicals:
1) Al: Aluminum
2) CuCl2: Copper Chloride
3) Cu(NO3)2: Copper Nitrate
4) (NH4)2SO4: Ammonium Sulfate
5) KMnO4: Potassium Permanganate
6) H2O2: Hydrogen Peroxide
1) Al: Aluminum
2) CuCl2: Copper Chloride
3) Cu(NO3)2: Copper Nitrate
4) (NH4)2SO4: Ammonium Sulfate
5) KMnO4: Potassium Permanganate
6) H2O2: Hydrogen Peroxide
Reactions:
1) Double Displacement: Cu(NO3)2 + (NH4)2SO4 ---> CuSO4 + 2NH4(NO3)
2) Single Displacement: Al + CuCl2 ---> Cu + AlCl2
3) Decomposition/Production of Gas: KMnO4 + H2O2 ---> O2 + H2O + MnO2 + K2O
1) Double Displacement: Cu(NO3)2 + (NH4)2SO4 ---> CuSO4 + 2NH4(NO3)
2) Single Displacement: Al + CuCl2 ---> Cu + AlCl2
3) Decomposition/Production of Gas: KMnO4 + H2O2 ---> O2 + H2O + MnO2 + K2O
Visual Evidence & documentation
Procedure:
Step 1 - A ball rolls down a 14 centimeter long inclined plane
Step 2 - The ball drops into a styrofoam cup; cup lowers under the added weight
Step 3 - Ammonium Sulfate and Copper Nitrate mingle in the cup
Step 4 - Double Displacement reaction creates a precipitate and colour change
Step 5 - Pulley system with an MA of 2 lowers the weighted cup
Step 6 - Cup tips lever with Copper Chloride
Step 7 - Copper Chloride splashes on a piece of Aluminum foil
Step 8 - The Single Replacement reaction dissolves the metal releasing a prepositioned ball
Step 9 - Ball rolls down a 20 centimeter long inclined copper pipe
Step 10 - Metal ball makes contact with electrical wires at the bottom end of the pipe
Step 11 - Ball completes the circuit turning on a mini Incandescent Light Bulb
Step 12 - The Demonstrator moves to a beaker of Hydrogen Peroxide separated from main project
Step 13 - The Demonstrator tips a vial of Potassium Permanganate into beaker
Step 14 - Decomposition Reaction occurs creating Steam and Oxygen Gas upon immediate contact
Step 1 - A ball rolls down a 14 centimeter long inclined plane
Step 2 - The ball drops into a styrofoam cup; cup lowers under the added weight
Step 3 - Ammonium Sulfate and Copper Nitrate mingle in the cup
Step 4 - Double Displacement reaction creates a precipitate and colour change
Step 5 - Pulley system with an MA of 2 lowers the weighted cup
Step 6 - Cup tips lever with Copper Chloride
Step 7 - Copper Chloride splashes on a piece of Aluminum foil
Step 8 - The Single Replacement reaction dissolves the metal releasing a prepositioned ball
Step 9 - Ball rolls down a 20 centimeter long inclined copper pipe
Step 10 - Metal ball makes contact with electrical wires at the bottom end of the pipe
Step 11 - Ball completes the circuit turning on a mini Incandescent Light Bulb
Step 12 - The Demonstrator moves to a beaker of Hydrogen Peroxide separated from main project
Step 13 - The Demonstrator tips a vial of Potassium Permanganate into beaker
Step 14 - Decomposition Reaction occurs creating Steam and Oxygen Gas upon immediate contact
Resolution
This project was a blend of all I have learned in this school semester of chemistry, but i decided to put it in this unit "Reactions & Chemical Bonding". Whilst creating this project, I realized that the core portion of this project leaned heavily toward the Chemical Reactions, and the study regarding them. The electrical circuitry, and physical building portions were not the main lesson objective. The purpose of this particular project was to focus on Chemical Reactions and the Stoichiometrics while also introducing other, more familiar materials to it. I thought this project was fun, and I was surprised by how dedicated I was to finishing this project. I stayed in on some days to work more on the project, and brought several items from my household instead of making do with school materials. I researched fun reactions that were complex, and very entertaining (I also made sure these reactions were safe enough). It's honestly the most amount of fun iv'e had in a school project for quite a while. This project has gotten me intrigued into more advanced chemistry as well as other fields like Pyrotechnic and Organic Chemistry.