HAND WARMER Project
This project was an intriguing project because of it's real world applications. The SMHS Soccer coaches had asked the STEM chemistry classes to design and produce hand warmers for the players. At our school, Soccer season was previously during the Autumn, but than changed to Winter. Because of this, the players experience extreme cold and discomfort during practice. It was our task to help the Soccer team and create cheap hand warmers that would increase in temperature by 20 degrees Celsius and hold the heat for an extended duration. After researching and testing the most effective strategies for hand warming, our group decided on using Calcium Chloride as a solute in an exothermic reaction. This reaction with water (H2O) and Calcium Chloride (CaCl2) was cheap, effective and if used in the right ratio, it could raise the temperature by exactly 20 degrees Celsius. We found that we could control the duration with the type of Calcium Chloride (coarse rock or fine dust). Also through careful testing and some calculations, we came up with a ratio of 5:35 for the reaction (5 mL of H2O : 35 g of CaCl2) to raise the surrounding temperature by precisely 20 degrees Celsius.
Thermochemistry: Branch of chemistry concerned with the amounts of heat energy evolved or absorbed during chemical reactions.
Calorimetry: In thermochemistry. It is the science of measuring heat in a physical and chemical process and observing absorbed or lost heat energy.
Enthalpy: Is the thermodynamic quantity of the total heat in a system; It is the total sum of internal energy and product of pressure and volume.
Entropy: A thermodynamic quantity that represents a system's increase in degrees of disorder or randomness. Entropy is commonly associated with Thermal Energy and it's correlation with Mechanical Energy.
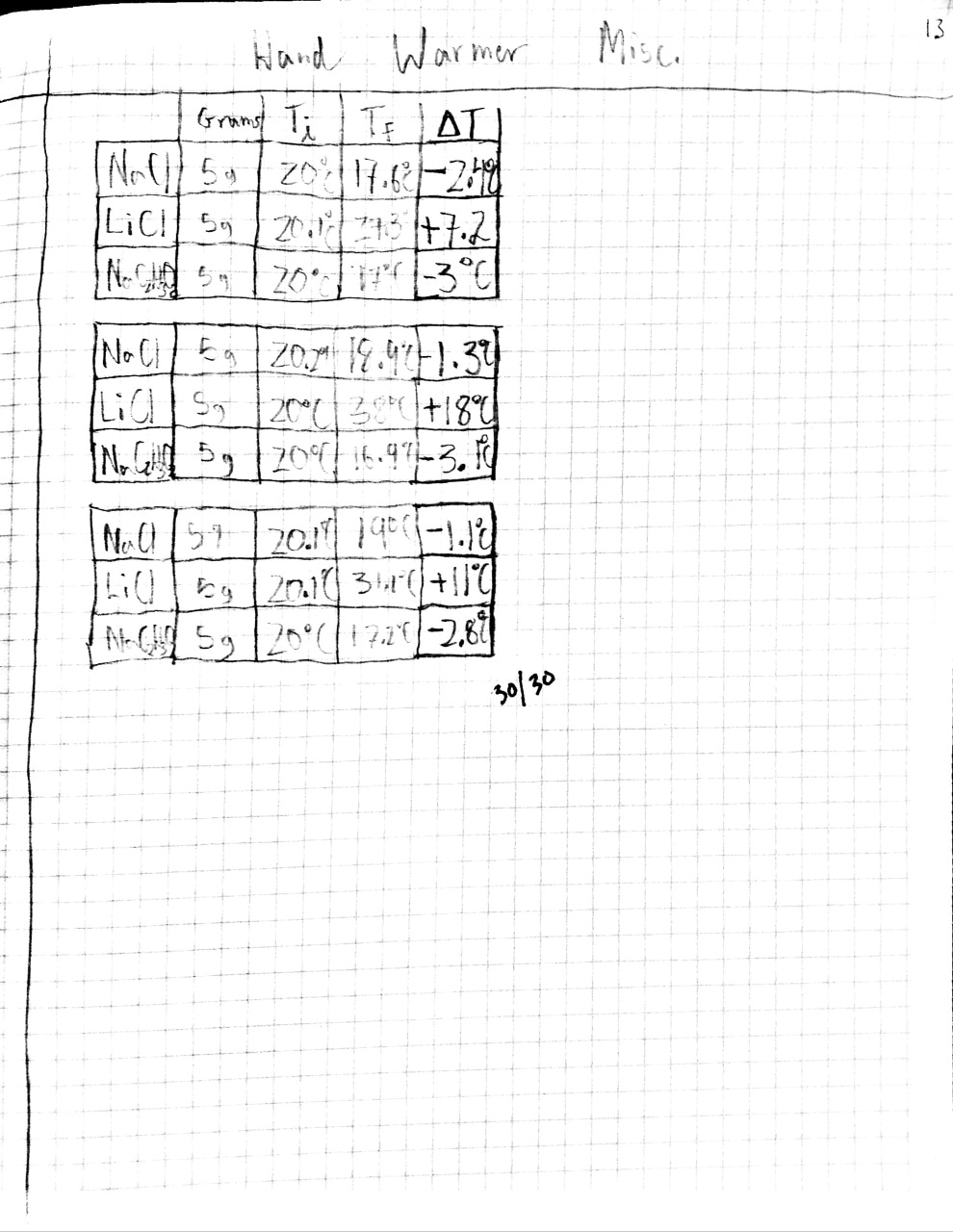
Endothermic: Reaction in witch heat energy is absorbed from the surroundings into the reactants/products. The surroundings lose heat while the reactants/products gain heat. These reactions are indicated with a loss in temperature. (Positive+ sign)
Exothermic: Reaction in witch heat is released from the reactants/products into the surroundings. The surroundings gain heat while the reactants/products lose heat. These reactions are indicated with a rise in temperature. (Negative - sign)
Disassociation: Processes in witch Ionic Compounds separate or split into smaller particulates or molecules to mingle together.
Ionic Compounds: Chemical Compounds composed of ions held together, electrically balanced (The Ionic Compound is neutral). A compound consists of an Anion and Cation or Polyatomic Anion and Polyatomic Cation.
Calorimetry: In thermochemistry. It is the science of measuring heat in a physical and chemical process and observing absorbed or lost heat energy.
Enthalpy: Is the thermodynamic quantity of the total heat in a system; It is the total sum of internal energy and product of pressure and volume.
Entropy: A thermodynamic quantity that represents a system's increase in degrees of disorder or randomness. Entropy is commonly associated with Thermal Energy and it's correlation with Mechanical Energy.
Endothermic: Reaction in witch heat energy is absorbed from the surroundings into the reactants/products. The surroundings lose heat while the reactants/products gain heat. These reactions are indicated with a loss in temperature. (Positive+ sign)
Exothermic: Reaction in witch heat is released from the reactants/products into the surroundings. The surroundings gain heat while the reactants/products lose heat. These reactions are indicated with a rise in temperature. (Negative - sign)
Disassociation: Processes in witch Ionic Compounds separate or split into smaller particulates or molecules to mingle together.
Ionic Compounds: Chemical Compounds composed of ions held together, electrically balanced (The Ionic Compound is neutral). A compound consists of an Anion and Cation or Polyatomic Anion and Polyatomic Cation.
HAND WARMER Design & Test Trials
4 Laws of Thermodynamics
0 Law): If two thermodynamic systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other.
1 Law): Energy can neither be created nor destroyed. It can only change forms. In any process, the total energy of the universe remains the same. For a thermodynamic cycle the net heat supplied to the system equals the net work done by the system.
2 Law): The entropy of an isolated system not in equilibrium will tend to increase over time, approaching a maximum value at equilibrium.
3 Law): As temperature approaches absolute zero, the entropy of a system approaches a constant minimum.
0 Law): If two thermodynamic systems are each in thermal equilibrium with a third system, then they are in thermal equilibrium with each other.
1 Law): Energy can neither be created nor destroyed. It can only change forms. In any process, the total energy of the universe remains the same. For a thermodynamic cycle the net heat supplied to the system equals the net work done by the system.
2 Law): The entropy of an isolated system not in equilibrium will tend to increase over time, approaching a maximum value at equilibrium.
3 Law): As temperature approaches absolute zero, the entropy of a system approaches a constant minimum.
Synopsis
This project was very interesting and fun. My group faced many problems, but we managed to discuss and solve each problems together. The greatest problems we faced were the calculations. When we were calculating how much grams of Calcium Chloride to use, we kept getting values that were way out of the expected range. After checking our calculations over and over again, we finally found our mistake in the conversion from "g/mol" to "g" at the end of the calculations. It was frustrating at first, but when we figured out our mistake, we were proud of ourselves. Another problems we faced were making the prototypes of our design. We faced problems of leakage; we made around five to ten hand warmers just because of leakage. We ended up adding double outer layers to prevent leakage. Also, we made sure to seal the bags thoroughly. There were few more minor problems we faced, but my group worked well together and we were satisfied with our product. We learned to work together by communicating and sharing ideas and ways to solve the problems. The chemistry part of this project was very hard to understand at first, but through trial and error and making mistakes in calculations, now we understand it very well. Overall, my group was satisfied with our hand warmer and the presentation.